
Data sources
A retrospective cohort study was carried out using two U.S. insurance claims databases: one with open claims and one with closed claims. Open claims are derived from multiple sources such as facilities and pharmacy systems and may include some healthcare interactions from each patient over an open-ended period [21]. Closed claims are derived directly from insurance providers [21] and show a comprehensive view of each patient’s healthcare interactions, but only during the patient’s enrollment period. The databases used in this study were:
-
1.
Symphony Health (SH): An open claims database including over 16 million patients with any claims based on diagnoses and procedures relating to pregnancy from January 2015 through December 2018 (Symphony Health, an ICON plc Company, PatientSource®).
-
2.
Myriad Genetics-Komodo Health (MGKH): A closed claims database that included pregnant patients who received a prenatal cell-free DNA screening test (Prequel) from Myriad Genetics, Inc. from September 2015 through January 2023, de-identified and linked to the Komodo Healthcare Map™ closed claims.
Both databases used in this study contained medical and pharmacy claims in addition to limited payer and demographic information. Of note, pharmacy claims are structured similarly in both databases despite being derived from different sources, though they vary in completeness based on the open or closed claims nature of the dataset [21]. Data linkage for the MGKH cohort was performed using tokenization to maintain patient privacy in Myriad’s and Komodo’s individual data sources (Datavant, Inc., San Francisco, CA) [22]. Symphony Health data was not linked to Myriad Genetics data. This study utilized de-identified data obtained in compliance with HIPAA regulations. Consequently, the study did not meet the U.S. Department of Health and Human Services’ definition of research involving human subjects (45 CFR 46.102). Therefore, it was not submitted for Institutional Review Board approval, nor did it require consent to participate. Additionally, the study adhered to the ethical principles outlined in the Declaration of Helsinki.
Study population
A cohort of individuals with PPD was assembled from each database. Exclusion and inclusion criteria differed slightly between the two cohorts based on the open vs. closed nature of the databases (Fig. 1). For both datasets, patients 18 years and older with an International Classification of Diseases 10th revision (ICD-10) code for outcome of delivery (Z37.xx) were identified between October 1, 2015 and December 31, 2017 for the SH cohort and between October 1, 2015 and January 31, 2022 for the MGKH cohort. Among these, patients with at least one ICD-10 code for depression (Table S1) between 5 and 180 days following the first delivery code, as well as a full 365 days of follow-up after the first depression diagnosis date (henceforth called the PPD diagnosis date), were included. Notably, the ICD-10 code (F53) was included; although it was labeled ‘Puerperal psychosis,’ it was intended to be used for both PPD and postpartum psychosis prior to October 2018. A similar approach, i.e., considering codes for both PPD and MDD used during the postpartum period as indicative of a PPD diagnosis, has been used by several other claims-based studies [15, 16, 23,24,25]. A buffer of 5 days after delivery was used to avoid capturing depression diagnoses coded during hospitalization for delivery; these codes may be referencing historical rather than ongoing conditions. In the MGKH cohort, patients were required to have at least 180 days of closed enrollment in medical and pharmacy coverage following delivery and at least 365 days of closed enrollment in pharmacy coverage following PPD diagnosis to ensure complete capture of diagnosis codes and medication fills. The study objective was to describe treatment patterns in all patients with PPD; therefore, patients with a history of depression or previous treatment for depression, who themselves have a higher risk of being diagnosed with PPD [24, 26], were included in the study.
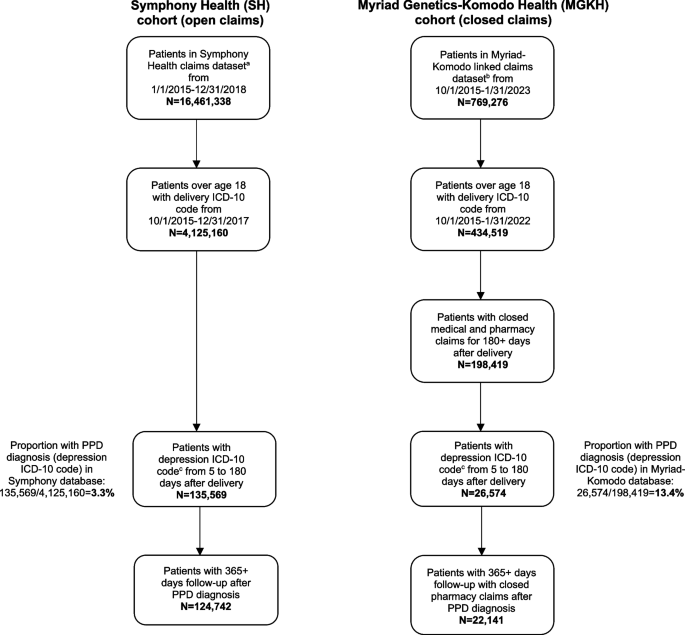
Cohort diagram. aSymphony Health claims dataset includes patients with any pregnancy-related claims from January 2015 through December 2018 (Symphony Health, an ICON plc Company, PatientSource®). bMyriad Genetics-Komodo Health claims dataset includes pregnant patients who received a prenatal cell-free DNA screening test (Prequel) from Myriad Genetics, Inc. from September 2015 through January 2023, de-identified and linked to the Komodo Healthcare Map™ closed claims. cDepression ICD-10 codes used to define PPD cases are listed in Table S1
Baseline characteristics were defined at the time of delivery, PPD diagnosis date, or using the closest available claims or enrollment data. Ethnicity data in the MGKH cohort was self-reported by patients and collected by Myriad Genetics, Inc. at the time the prenatal test was ordered. The frequency of psychiatric medication fills during the presumed pregnancy period (280 days before delivery) and pre-pregnancy period (560 to 281 days before delivery) was assessed in the SH cohort as a proxy for prior mental health treatment. The frequency of diagnoses for anxiety (ICD-10 codes F41.xx), bipolar disorder (ICD-10 codes F31.xx), and schizophrenia/schizoaffective disorder (ICD-10 codes F20.xx, F25.xx) during the study period (5 to 180 days after delivery) was also assessed.
Identification of pharmacotherapy treatment patterns
Psychiatric medication fills were identified for all patients in the 365 days following the PPD diagnosis. The 56 medications of interest (Table S2) included antidepressants (selective serotonin reuptake inhibitors [SSRIs], serotonin/norepinephrine reuptake inhibitors [SNRIs], serotonin antagonist and reuptake inhibitors [SARIs], bupropion, monoamine oxidase inhibitors [MAOIs], tricyclics, novel, and alpha-2 antagonists), antipsychotics (typical and atypical), and select mood stabilizers (antiseizure agents and lithium). The drug generic name, start date, and days of supply were captured for each fill.
Treatment patterns in the 365 days following PPD diagnosis were described by the number of unique psychiatric medications filled and the number of treatment lines. The number of treatment lines was defined as the number of times any new medication(s) was(were) filled during the study period, regardless of whether previous medications were continued. For example, if a patient filled one medication on the day of diagnosis and another two medications were later filled on the same date one month later, this was considered two distinct treatment lines and three distinct medications. The time in days from PPD diagnosis to the first observed fill in the study period and the proportion of patients who filled only one prescription were also reported. The top ten most common medications and medication classes were summarized by treatment line up to the third treatment line following diagnosis. Treatment lines were numbered based on observation within the study period; as such, a ‘first-line’ treatment in this study may have previously been prescribed to that patient before the study period began.
Additionally, continuous-treatment episodes were determined using the AdhereR package [27] applied in two ways: 1) applied to all psychiatric medications together to define each patient’s continuous-treatment episodes on any medication, which was used to estimate the length of pharmacotherapy and the timing and frequency of discontinuation, and 2) applied to each psychiatric medication individually to define each patient’s continuous-treatment episodes for each specific medication filled, which was used to characterize the frequency of different medication combinations. A continuous-treatment episode was defined based on the timing of fills as well as the days of supply of each fill, allowing for gaps of ≤30 days between when the previous medication supply would have run out and the next medication was filled [17]. Medication supply was allowed to carry over within a given drug, so that if a prescription was refilled before the previous supply ran out, the refilled drug was assumed to have been started once the previous supply was finished. Discontinuation was defined by the presence of any gap in treatment episodes > 30 days, while medication combinations were defined as periods where two or more medication-specific treatment episodes overlapped for ≥30 days.
Statistical analysis
This was a descriptive study; no statistical testing was performed. The analyses were performed using R Statistical Software (version 4.3.1; R Core Team 2023; R Foundation for Statistical Computing, Vienna, Austria; URL:
link





